From clear to purple or brown, that’s how irradiation runs
by Ken Previtali
01 December 2013
Regarding the post on “Artificially Purpled Glass” with the link to David Whitten’s fantastic website:
David also mentions the effects radiation has on bottles containing selenium. “Clear glass that was originally decolorized by using selenium as the active ingredient instead of manganese (the use of selenium was particular prevalent in the 1920s and 1930s and later) may turn strange shades of “burnt amber” or “smoky dull dark brown” under irradiation. . .”

Here’s an Art Deco example from the late 20s or early 30s made by the F. E. Reed Glass Company, Rochester, NY (ginger ale, of course). Green Mountain Ginger Ale Company, Manchester, NH (a noteworthy location in July 2013). The irradiated bottle (on right) was a gift from Carl Pratt a few years ago.
In his article, David describes the un-articifical way clear glass will turn purple: “Glass containing enough manganese may naturally slowly turn some shade of very light to medium purple after several years of exposure to the rays of the sun.” Bottles with selenium content from the same period can have a subtle overall tint in a light yellowish brown or “straw” color, which I believe is a result of the same natural process David describes for “sun-colored amethyst”. They are not very common (or perhaps overlooked), and I don’t know how long it takes for the chemical change to take place. Anyone for “sun-colored straw”?
A long time southern collector I knew for many years (now passed on), was fond of selling irradiated bottles. He was very strict in labeling the bottles and clearly telling buyers that the bottles had been altered and were not the original color. I asked him one day how he turned his bottles. Here’s the story pretty much as he told it:
“I live near a poultry processing plant and the manager was a friend of mine. Since I knew that chickens were routinely irradiated I asked him if he could “zap” my bottles, and he said sure bring ’em on down. I brought about ten boxes of ones I thought would turn. He told me ‘Set the boxes here at the head of the conveyor line and my guys will plop ’em on the belt. You go way down to the other end of the plant and pick ’em off the line when they come through.’ Well, with my white visitor’s coat on, I went down as he said and waited. There were a couple of fellas there watching the production line. I said nothing, and I guess they figured I was an inspector or something. It was chicken, chicken, chicken, chicken, chicken on the belt for a while. Then came chicken, chicken, box, chicken, chicken, box, chicken, chicken, box and so on. I pulled them off as they came through. The fellas didn’t know what to make of it as their boss hadn’t told them. The look on their faces when those boxes came through! I told them what was going on, and gave each a nice purple bottle when it was all over.”
From clear to purple or brown that’s how irradiation runs. . . whether by machine or by sun.

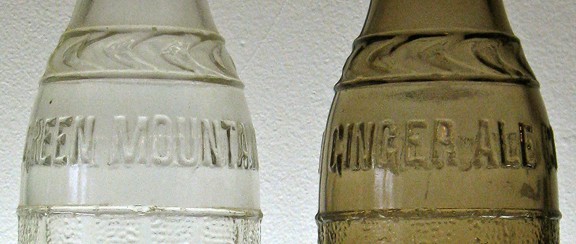






The rest of the story or so I was once told, is how one can TURN THEM BACK to their original color. Supposedly sun-colored bottles, when heated up, will return to their original color. I do not know at what temperature or for how long or if it is even true. Nor do I know if this applies to irradiated bottles. In any case, I have never tried to do this, wanting to keep the purple in the few SCA pieces I’ve had. Half-baked idea?
Froggy- you’ll find it in the Joy of Cooking: “In a 300 degree F oven, bake for approximately for several hours, or until clear color appears uniformly.” See? You are not half-baked!
Your post got me wondering, as I had not heard that irradiated bottles, sun- or gamma ray- induced could be reversed. What you were told was correct and the bogus cookbook “recipe” above is actually what I found in a few scientific journals. In machine-irradiated bottles the atoms of silicon in the glass are “knocked about” by the high-energy gamma rays, changing how visible light can travel through the glass. Depending on the chemicals in the glass, this change enables specific colors of light to be absorbed and then visible to us, appearing as the color of the glass. Heating the glass, essentially “annealing” it, shakes up the atoms enough to allow the displaced silicon atoms to find their way home, and the glass turns clear again. I could not find any mention of whether this reversal would work on SCA or SCS (Sun-colored straw) bottles. Maybe an intrepid collector will risk a sun bottle and perhaps their oven as well to complete the rest of the story you added. Thanks for the comment.